Adaptive cryo-EM reconstruction: a review of current mouse models for muscular dystrophies. Curr. Opin. 12, 349-361 (2018)
Punjani, A., Zhang, H., and Fleet, D.J. discuss adaptive regularization and single-particle cryo-EM reconstruction. Nat. Methods 17, 1214–1221 (2020).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
A review of current mouse models of muscular dystrophies. Curr. Opin. It’s named Genet. There was a Dev. 12, 349–361.
Jung, D., Yang, B., Meyer, J., Chamberlain, J. S. & Campbell, K. P. Identification and characterization of the dystrophin anchoring site on beta-dystroglycan. J. Biol. Chem. 270 was published in 1995.
The impact on the mucin-like region of -dy of -O-GalNAc and -O-Man glycan protein modifications was compared. Glycobiology 31, 649–661, was published in 2011.
Esampa, C. T., Bentham, G.R., Kroger, S. and D. J. The effects of post-translational processing on dystroglycan synthesis and trafficking. FEBS Lett. 555, 209–216 (2003).
Duchenne muscular dystrophy and dystroglycy. A Pediatr. The patient with primary adhalinopathy is a cell
Duchenne and Becker muscular dystrophy genetics and emerging treatments. Pediatr. It was Clin. North. Am. 62, 723–742 (2015).
Muthu, M., Richardson, K. A. & Sutherland-Smith, A. J. The crystal structures of dystrophin and utrophin spectrin repeats: implications for domain boundaries. PLoS ONE 7, e40066 (2012).
A paper by Ramaswamy and K. S. Lateral transmission of force is impaired in skeletal muscles of dystrophic mice and very old rats. The J. Physiol. 589 was published in 2011.
Guo, C. et al. Duchenne muscular dystrophy was caused by myopathy caused by 7 integrin in dystrophin- deficient mice. Hum. The name isMol. A person who is related to another person. 15, 989–998 (2006).
G. et al. had a paper. Sarcolemmal alpha and gamma sarcoglycan protein deficiencies in Turkish siblings with a novel missense mutation in the alpha sarcoglycan gene. It is a Pediatr. There was a neurological disease. There were 50 articles in the year of 50.
Piccolo, F. et al. The cause of primary adhalinopathy is muscular-dystrophy. Nat. Genet. There was a story about 10, 243–245 in 1995.
Saha, M. et al. There are correlations with genetic analyses of limb-girdle muscular dystrophy in Saudi Arabia and Sudan. Physiol. Genomics 50, 929–939 (2018).
Piccolo, F. et al. A founder mutation in the γ-sarcoglycan gene of Gypsies possibly predating their migration out of India. Hum. Mol. The person is named Genet. May 5, 1996
Dai, Y. et al. Whole exome sequencing identified a novel DAG1 mutation in a patient with rare, mild and late age of onset muscular dystrophy-dystroglycanopathy. A cell. There is a word that refers to this. Med. 23 was published in 2019.
There are certain alterations in the dystrophin gene that are associated with dilated cardiomyopathy. There is a book called “Mol. Genet. Metab. 77, 119–126 (2002).
Cadherin domains in the polysaccharide degrading marine bacterium Saccharophagus degradans 2-40 are carbohydrate-binding modules
Fraiberg, M., Borovok, I., Bayer, E. A., Weiner, R. M. & Lamed, R. Cadherin domains in the polysaccharide-degrading marine bacterium Saccharophagus degradans 2-40 are carbohydrate-binding modules. J. Bacteriol. 193, 283–285 (2011).
Vickers, C. et al. Endo-fucoidan hydrolases from glycoside hydrolase family 107 (GH107) display structural and mechanistic similarities to α-l-fucosidases from GH29. J. Biol. In Chem. 293, 18796 and 18308 this year.
It was shown that autoproteolytic coupled to the folding of theprotein in the SEA domain of theMUC1 mucin could be achieved. Struct. The journalMol. Biol. 13, 70–78, was published in 2006).
The sarcoglycan–Sarcospan subcomplex was analyzed. Exp. Cell. Res. 313 was published in 2007.
The structure of the polysialic acid degrading endosialidase of K1F was crystalized. There was a structuring. Mol. Biol. 12, 90–96 (2005).
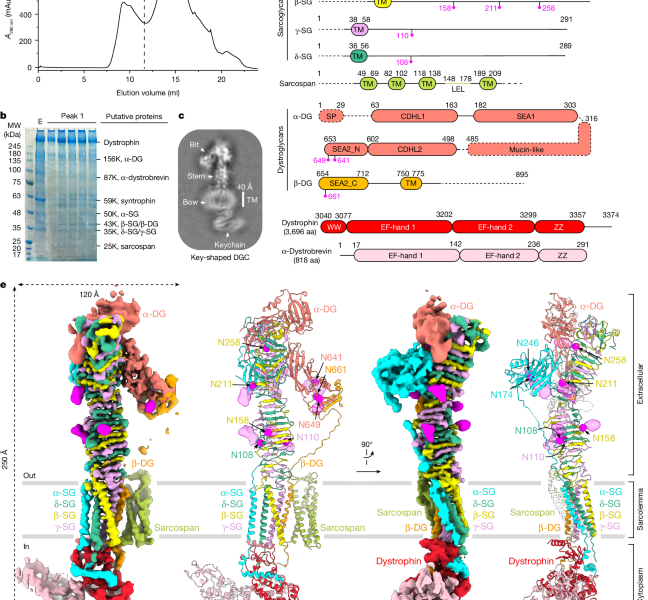
Source: Native DGC structure rationalizes muscular dystrophy-causing mutations
The genetic mechanism of -dystrobrevin in skeletal muscle and its neuromuscular junctions: a review by Marshall, J. M. et al
R. M. et al. Tyrosine-phosphorylated and nonphosphorylated isoforms of α-dystrobrevin: roles in skeletal muscle and its neuromuscular and myotendinous junctions. The J. Cell Biol. 160 was published in the year 2003
Grady, R. M. et al. There is genetic evidence for the roles of the dystrophicin–glycoprotein complex. Neuron 25, 279–293 (2000).
Ponting, C. P., Blake, D. J., Davies, K. E., Kendrick-Jones, J. & Winder, S. J. ZZ and TAZ: new putative zinc fingers in dystrophin and other proteins. Trends Biochem. The journal sci 21, 11–9 and 13 were written in 1996.
Marshall, J. L. et al. Muscular regeneration and Utrophin expression require a sarcospan-dependent Akt activation. The cell system is referred to as J. Cell Biol. The film is titled “S197, 1009–1 027”.
Source: Native DGC structure rationalizes muscular dystrophy-causing mutations
Partner-protein recognition and structure: How dystroglycan governs early embryonic development and is responsible for triggering sarcoglycanopathy
The principles of partner-protein recognition and their interplay with structure are discussed by Susa, Kruse and Blacklow. Trends Cell. https://doi.org/10.1016/j.tcb.2023.09.003 (2023).
Williamson, R. A. et al. Early embryonic development can be disrupted by dystroglycan in mice. I don’t know what to say. Mol. Genet. There were 6, 833 in 1997.
Miller, G., Peter, A. K., Espinoza, E., Heighway, J. & Crosbie, R. H. Over-expression of Microspan, a novel component of the sarcoplasmic reticulum, causes severe muscle pathology with triad abnormalities. There is a book called J. Muscle Res. Cell Motil. 27 was published in 2006
Yoshida, M. et al. Biochemical evidence for association of dystrobrevin with the sarcoglycan–sarcospan complex as a basis for understanding sarcoglycanopathy. Hum. Mol. It is a Genet. 9, 1033–1040 (2000).
Source: Native DGC structure rationalizes muscular dystrophy-causing mutations
Crystal structure of rhamnogalacturonase A from Aspergillus aculeatus: A new domain of virulence factors
The structure of a plant virulence factor is new to the domain. Science 260, 1503–1507 (1993).
P., Charles, I., Fairweather, N. F., and N. W. analyzed the structure of Bordetella pertussis virulence factors. Nature 381, 90– 92 was published in 1996.
Petersen, T. N., Kauppinen, S. & Larsen, S. The crystal structure of rhamnogalacturonase A from Aspergillus aculeatus: a right-handed parallel β helix. Structure 5, 533–544 (1997).
Parvatiyar, M. S. and others. The regulates the cardiac isoproterenol response. J. Am. The heart is the main organ of the body. 4, e002481 (2015).
Mitchell, R. D., Palade, P. & Fleischer, S. Purification of morphologically intact triad structures from skeletal muscle. J. Cell Biol. 96, 1008–1016 (1983).
Some new tools are available for automated single-particle analysis. J. 478, 4169–4185 (2021).
Bepler, T. et al. Positive-unlabeled convolutional neural networks for particle picking in cryo-electron micrographs. Nat. Methods 16, 1153–1160 (2019).
The optimal determination of particle orientation, absolute hand, and contrast loss can be determined using single-particle electron cryomicroscopy. J. Mol. Biol. 333, 721–745 (2003).
Source: Native DGC structure rationalizes muscular dystrophy-causing mutations
Predicting the structure of interactions between biomolecular systems: an all-atom proof of the molecule-by-molecule approach to molecular crystallography
J. Abramson, et al. Predicting the structure of interactions between biomolecules. The nature story was published in the journal Nature and can be found here.
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. A. Biol. The Crystallogr. 66 was published in 2010.